Research Areas in the Roberts Laboratory

ROBERTS GROUP
The vast majority of biopharmaceutical products (as opposed to “small-molecule” drugs such as tablets and capsules) that are available commercially or are in late-stage clinical testing are based on one or more proteins as the active ingredient. Biopharmaceuticals are also the fastest-growing sector of the pharmaceutical industry, and target diseases ranging from diabetes and cancer to auto-immune disorders. Many of the research projects in the Roberts laboratory are motivated by or directly focused on topics that fall within the general topic of protein stability and assembly within the context of biotechnology projects.
Current Group Members: Lily Motobar, Terrance Shoemaker, James Forder, Jordan Berger, Hassan Shahfar, Veerabhadraiah Palakollu
Protein-protein and protein-solute interactions
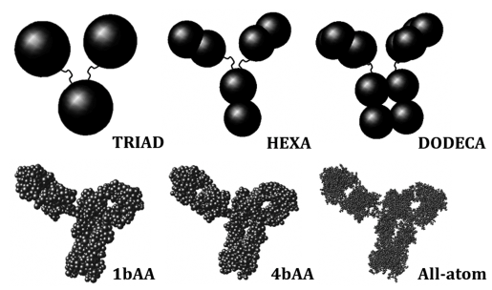 Proteins interact continuously with the surrounding solvent, other proteins, and (co)solutes in solution. Those interactions span from strong “binding” events to weaker interactions. When considering weaker interactions, averaged over the entire ensemble of protein configurations and surrounding molecular environment, these tend to strongly influence protein phase behavior, separation processes, and protein aggregation rates. Research in this area focuses on improved experimental and theoretical ways to quantify the relevant interactions, as well as how these change as one moves across a broad range of solution conditions that are needed for manufacturing and application of biotechnology products. This work is supported by funding from the NSF, NIH, NIST and a number of biopharmaceutical companies.
Proteins interact continuously with the surrounding solvent, other proteins, and (co)solutes in solution. Those interactions span from strong “binding” events to weaker interactions. When considering weaker interactions, averaged over the entire ensemble of protein configurations and surrounding molecular environment, these tend to strongly influence protein phase behavior, separation processes, and protein aggregation rates. Research in this area focuses on improved experimental and theoretical ways to quantify the relevant interactions, as well as how these change as one moves across a broad range of solution conditions that are needed for manufacturing and application of biotechnology products. This work is supported by funding from the NSF, NIH, NIST and a number of biopharmaceutical companies.
Recent Publications:
- Shahfar H, Du Q, Parupudi A, Shan L, Esfandiary R, Roberts CJ*, Electrostatically driven protein-protein interactions: quantitative prediction of second osmotic virial coefficients to aid antibody design, J Phys Chem Lett, 13:1366-1372 (2022)
- Shahfar H**, Forder J**, Roberts CJ*, Towards a suite of coarse-grained models for molecular simulation of monoclonal antibodies and therapeutic proteins, J Phys Chem B, 125:3574-3588 (2021)
- Pathak JA, Nugent S, Bender M, Roberts CJ, Curtis RJ, Douglas JF*, Comparison of Huggins coefficients and osmotic second virial coefficients of buffered solutions of monoclonal antibodies, Polymers, 13:601 (2021)
- Woldeyes MA, Qi W, Razinkov V, Furst EM*, Roberts CJ*, Temperature-dependence of protein solution viscosity and protein-protein interactions: Insights for the origins of high-viscosity protein solutions, Mol Pharm, 17:4473-4482 (2020)
- Ferreira GM, Shahfar H, Remmele RL, Sathish H, Roberts CJ*, Identifying key amino acids that drive electrostatic attractions between therapeutic antibodies, J Phys Chem B , 123:10642-10653 (2019)
- Woldeyes MA, Calero-Rubio C, Furst EM, Roberts CJ*, Light scattering to quantify protein-protein interactions at high protein concentrations, Methods Mol Biol, 2039:23-37 (2019)
- Calero-Rubio C, Saluja A, Sahin E, Roberts CJ*, Predicting high-concentration interactions of monoclonal antibody solutions: Comparison of theoretical approaches for strongly attractive vs. repulsive conditions, J Phys Chem B, 123:5709-5720 (2019)
- Ferreira GM, Calero-Rubio C, Remmele RL, Samra H, Roberts CJ, Electrostatically Mediated Protein-Protein Interactions for Monoclonal Antibodies: a Combined Experimental and Coarse-Grained Molecular Modeling Approach, J Pharm Sci, 108:120-132 (2019)
- Woldeyes MA, Lam LJ, Leiske D, Galush W, Roberts CJ, Furst EM, Viscosities and protein interactions of bispecific antibodies and their monospecific mixtures, Mol Pharm, 15:4745-4755 (2018)
- Woldeyes MA, Battistoni C, Razinkov V, Furst EM*, Roberts CJ*, How well do electrostatic protein-protein interactions predict changes in viscosity of monoclonal antibody solutions?, J Pharm Sci, 108:142-154 (2019)
- Yang D, Correia JJ, Stafford WF, Roberts CJ, Singh S, Hayes D, Kroe-Barrett R, Nixon A, Laue TM, Weak IgG self- and hetero-association characterized by fluorescence analytical ultracentrifugation, Protein Sci, 27: 1334-1348 (2018)
- O’Brien CJ, Calero-Rubio C, Razinkov V, Robinson AS, Roberts CJ, Biophysical characterization and molecular simulation of electrostatically driven self-association of an anti-fluorescein single-chain antibody, Protein Sci, 27:1275-1285 (2018)
- Calero-Rubio C, Saluja A, Ghosh R, Roberts CJ, Predicting high-concentration monoclonal antibody interactions with dilute solution data and coarse-grained molecular simulations, J Pharm Sci, 107:1269-1281 (2018)
- Calero-Rubio C, Strab C, Barnett GV, Roberts CJ, Protein partial molar volumes in multi-component solutions from the perspective of inverse Kirkwood-Buff theory, J Phys Chem B, 121:5897-5907 (2017)
- Woldeyes MA, Calero-Rubio C, Furst EM, Roberts CJ, Predicting protein interactions in concentrated globular protein solutions using colloidal models, J Phys Chem B, 121:4756-4767 (2017)
- Calero-Rubio C, Saluja A, Roberts CJ*, Coarse-grained antibody models for “weak” protein-protein interactions from low to high concentrations, J Phys Chem B, 120:6592-6605 (2016)
- Barnett GV, Razinkov V, Kerwin BA, Curtis RA, Roberts CJ*, Osmolyte Effects on Monoclonal Antibody Stability and Concentration-Dependent Protein Interactions with Water and Common Osmolytes, J Phys Chem B, 120:3318-3330 (2016)
- Barnett GV, Razinkov V, Kerwin BA, Hillsley A, Roberts CJ*, Acetate and Citrate Specific-Ion-Effects on Temperature-Dependent Aggregation Rates of Anti-Streptavidin IgG1, J Pharm Sci, 105:1066-1073 (2016)
- Barnett GV, Razinkov V, Kerwin BA, Laue TM, Woodka A, Butler PD, Perevozchikova T, Roberts CJ*, Specific ion effects on the aggregation mechanisms and protein-protein interactions for anti-streptavidin immunoglobulin gamma 1, J Phys Chem B, 119:5793-5804 (2015)
- Roberts CJ,* Blanco MA, Role of anisotropic interactions for proteins and patchy nanoparticles, J Phys Chem B, 118:12599-12611 (2014)
- Blanco MA, Martorana E, Manno M, Perevozchikova T, Roberts CJ*, Protein-protein interactions in dilute to concentrated solutions: alpha-chymotrypsinogen at acidic pH, J Phys Chem B, 118: 5817-5831 (2014)
- Blanco MA, Sahin E, Robinson AS, Roberts CJ*, Coarse-grained model of protein-protein interactions, B22, and protein cluster formation, J Phys Chem B, 117: 16013-16028 (2013)
- Grünberger A, Lai P-K, Blanco MA, Roberts CJ*, Coarse-grained modeling of protein second osmotic virial coefficients: sterics and non-specific attractions, J Phys Chem B, 117:763-770 (2013)
- Siderius DW*, Krekelberg WP, Roberts CJ, Shen VK, Osmotic virial coefficients for model protein and colloidal solutions: Importance of ensemble constraints, J Chem Phys, 136(17) 175102/1-9 (2012)
- Roberts CJ*, Das TK, Sahin E, Predicting, Aggregation Rates for Therapeutic Proteins: Approaches and Challenges, Int J Pharm, 418:318-333 (2011)
- Blanco MA, Sahin E, Li Y, Roberts CJ*, Reexamining Protein-Protein and Protein-Solvent Interactions from Kirkwood-Buff Analysis of Light Scattering in Multi-Component Solutions, J Chem Phys, 134:225103/1-12 (2011)
Protein (mis)folding, aggregation, and phase behavior
Proteins are heteropolymers of amino acids that typically must be properly folded in order to perform their biological function or to be useful as biotechnology products. An ever growing body of experimental evidence shows that correct folding and function is in competition with “off pathway” protein-protein interactions that result in misfolded or non-native aggregates composed of multiple protein chains. Although the exact role of such aggregates in the progression of neurodegenerative diseases remains unclear, insoluble aggregates are clearly causative agents for diseases such as cataracts and amyloidoses. More important from the perspective of pharmaceutical products, soluble or insoluble aggregates are a major risk factor for unwanted patient immune responses and safety issues with some protein-based products.
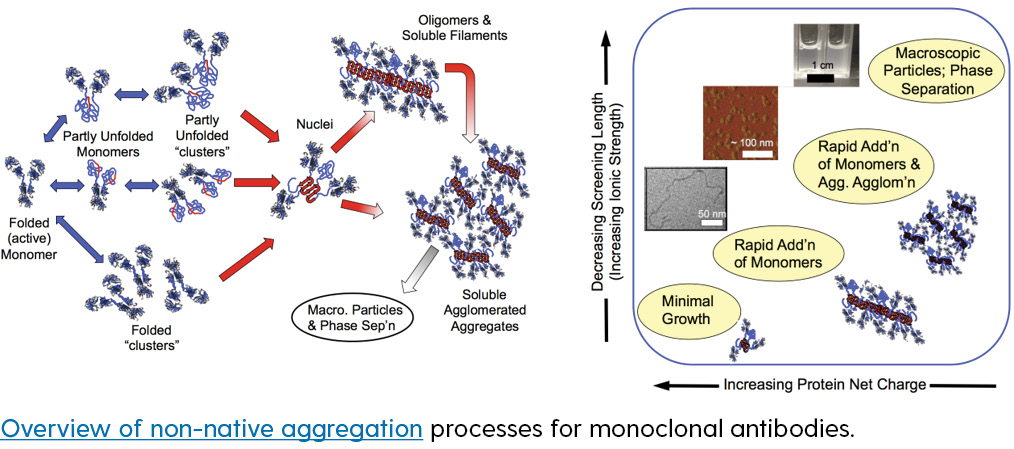
Research in this area focuses on elucidating the key mechanistic stages for aggregation of a range of different proteins, so as to gain insights regarding how to change product formulation or manufacturing conditions to balance the different stages of aggregation when one is seeking to maximize shelf life and/or control the morphology and size of the resulting aggregates. This involves a variety of experimental methods, as well as kinetic and thermodynamic modeling. This work is supported by funding from the NIH, NIST, and a number of biopharmaceutical companies.
Recent Publications:
- Gomes DC, Teixeira SCM, Leao JB, Butler P, Razinkov VI, Qi W, Rodriguez MA, Roberts CJ*, In situ monitoring of protein unfolding / conformational states under cold, high-pressure stress, Mol Pharm, 18:4415-4427 (2021)
- Meric G, Naik S, Hunter AK, Robinson AS*, Roberts CJ*, Challenges for design of aggregation-resistant variants of granulocyte-colony stimulating factor, Biophys Chem, 277:106630 (2021)
- Wood CV, Qi W, Razinkov V, Furst EM*, Roberts CJ*, A rapid, small-volume approach for protein aggregation via air-water Interfaces, J Pharm Sci, 110:1083-1092 (2021)
- Wood CV, Razinkov V, Qi W, Furst EM*, Roberts CJ*, Kinetics and competing mechanisms of antibody aggregation via bulk solution and surface-mediated pathways, J Pharm Sci, 109:1449-1459 (2020)
- Wang W, Roberts CJ, Protein Aggregation – Mechanisms, Detection, and Control, Int J Pharm Sci, 550:251-268 (2018)
- Gomes D, Kalman RK, Pagels R, Rodrigues MA, Roberts CJ*, Parallel Chromatography and In Situ Scattering to Interrogate Competing Protein Aggregation Pathways, Protein Sci, 27:1325-1333 (2018)
- Yang D, Correia JJ, Stafford WF, Roberts CJ, Singh S, Hayes D, Kroe-Barrett R, Nixon A, Laue TM, Weak IgG self- and hetero-association characterized by fluorescence analytical ultracentrifugation, Protein Sci, 27:1334-1348 (2018)
- Yang D, Kroe-Barrett R, Singh S, Roberts CJ, Laue TM*, IgG cooperativity – is there allostery? Implications in therapeutic antibody development, mAbs, 9:1231-1252 (2017)
- Rosa MF, Roberts CJ, Rodriguez MA*, Connecting High-Temperature and Low-Temperature Protein Stability and Aggregation, PLOS One, (online only) DOI: 10/1371/journal.pone.0176748 (2017)
- Meric UG, Robinson AS*, Roberts CJ*, Driving forces for non-native protein aggregation and approaches for predicting aggregation-prone regions, Ann Rev Chem Biomol Eng, 8 (2017), DOI: 10.1146/annurev-chembioeng-060816-101404
- Barnett GV, Drenski MC, Razinkov V, Reed WF*, Roberts CJ*, Protein aggregation mechanisms and kinetics from combined monomer depletion rates and continuous scattering, Analytical Biochem, 511:80-91 (2016)
- Barnett GV, Razinkov V, Kerwin BA, Hillsley A, Roberts CJ*, Acetate and Citrate Specific-Ion-Effects on Temperature-Dependent Aggregation Rates of Anti-Streptavidin IgG1, J Pharm Sci, 105:1066-1073 (2016)
- Wu H, Truncali K, Ritchie J, Kroe-Barrett R, Singh S, Robinson AS, Roberts CJ*, Weak protein interactions and pH- and temperature-dependent aggregation of human Fc1, mAbs, 7:1-12 (2015)
- Barnett GV, Razinkov V, Kerwin BA, Laue TM, Woodka A, Butler PD, Perevozchikova T, Roberts CJ*, Specific ion effects on the aggregation mechanisms and protein-protein interactions for anti-streptavidin immunoglobulin gamma 1, J Phys Chem B, 119:5793-5804 (2015)
- Amin S,* Barnett GV, Pathak J,* Roberts CJ, Sarangapani PS, Protein Aggregation, Particle Formation, Characterization, and Rheology, Curr Opin Coll Int Sci, 19:439-449 (2014)
- Roberts CJ, Protein Aggregation and Its Impact on Product Quality, Curr Opin Biotech, 30:211-217 (2014)
- Roberts CJ, Therapeutic protein aggregation: mechanisms, design, and control, Trends Biotech, 32: 372-380 (2014)
- Wu H, Kroe-Barrett R, Singh S, Robinson AS, Roberts CJ*, Competing aggregation pathways for monoclonal antibodies, FEBS Letters, 588: 936-941(2014)
- Costanzo JA, O’Brien CJ, Tiller K, Tamargo E, Robinson AS, Roberts CJ*, Fernandez EJ*, Computational Design to Control Protein Aggregation Rates Through Conformational Stability, Protein Engineering Design & Selection, 27: 157-167 (2014)
- Maurer RW, Hunter AK, Robinson AS*, Roberts CJ*, Aggregates of α-chymotrypsinogen anneal to access more stable aggregate states, Biotechnol Bioeng, 111: 782-791 (2014)
- Weiss WF IV, Zhang A, Jordan JL, Ivanova MA, Sahin E, Fernandez EJ, Roberts CJ*, Reduction of the C191-C220 disulfide of α-chymotrypsinogen A accelerates amyloid formation via reduced nucleation barriers, Biophys Chem, 185:79-87 (2014)
- Maurer RW, Hunter AK, Wang X, Wang WK, Robinson AS*, Roberts CJ*, Folding and aggregation of a multi-domain engineered immunotoxin, Biochem Eng J., 81:8-14 (2013)
- Murphy RM*, Roberts CJ., Protein misfolding and aggregation research: some thoughts on improving quality and utility, Biotechnol Prog, 29:1109-1115 (2013)
- Roberts CJ*, Nesta DP, Kim N., Effects of temperature and osmolytes on parallel degradation routes for an IgG1 antibody, J Pharm Sci, 102:3556-3566 (2013)
- Wang W*, Roberts CJ*, Non-Arrhenius Protein Aggregation, AAPS J, (online only) doi:10.1208/s12248-013-9485-3 (2013)
- Kim N, Remmele RL, Liu D, Razinkhov V, Fernandez EJ, Roberts CJ*, Aggregation of Anti-Streptavidin Immunoglobulin Gamma-1 Mediated by Fab Unfolding and Competing Growth Pathways, Biophys Chem, 172:26-36 (2013)
- Sahin E, Roberts CJ*, Size-Exclusion Chromatography with Multi-Angle Light Scattering (SEC-MALS) for Elucidating Protein Aggregation Mechanisms, Methods Mol Biol, 899:403-423 (2012)
- Kroetsch AM, Sahin E, Wang H-Y, Krizman S, Roberts CJ*, Relating particle formation to salt- and pH-dependent phase separation of non-native aggregates of alpha-chymotrypsinogen A, J Pharm Sci, 101:3651-3660 (2012)
- Sahin E, Weiss WF IV, Kroetsch AM, King KR, Kessler RK, Das TK, Roberts CJ*, Non-native aggregation and pH-temperature phase behavior of aggregates for an IgG2 antibody, J Pharm Sci, 101:1678–1687 (2012)
- Roberts CJ*, Das TK, Sahin E, Predicting Aggregation Rates for Therapeutic Proteins: Approaches and Challenges, Int J Pharm, 418:318-333 (2011)
- Brummitt RK, Nesta DP, Chang L, Chase SF, Laue TM, Roberts CJ*, Non-native aggregation of an IgG1 antibody in acidic conditions: 1. Unfolding, colloidal interactions, and formation of amyloid-like high molecular weight aggregates, J Pharm Sci, 100:2087-2103 (2011)
- Brummitt RK, Nesta DP, Chang L, Kroetsch AM, Roberts CJ*, Non-native aggregation of an IgG1 antibody in acidic conditions: 2. Nucleation-and-growth kinetics with competing growth mechanisms, J Pharm Sci, 100:2104-2119 (2011)
- Sahin E, Jordan JL, Zhang A, Naranjo A, Costanzo JA, Weiss WF IV, Spatara ML, Robinson AS, Fernandez EJ*, Roberts CJ*, Computational design and biophysical characterization of point mutations for gamma-D-crystallin illustrate a balance of conformational stability and intrinsic aggregation propensity, Biochemistry, 50:628-639 (2011)
Protein interactions, aggregation, and viscosity at high concentrations
Protein interactions and stability can be dramatically altered by moving from traditional “low” protein-concentration conditions (< approx.. 10 g/L) to “high” concentrations (~ 150+ g/L). Working at high concentrations can alter unfolding free energies, increase viscosity to unmanageable levels, induce phase separation, and greatly accelerate aggregation. Research in our group focuses on development of improved experimental and theoretical methods to quantify and predict how protein interactions and the resulting product properties are altered by moving from low to high protein concentrations. This work is supported by funding from the NIH, NIST and a number of biopharmaceutical companies.
Recent Publications:
- Shahfar H**, Forder J**, Roberts CJ*, Towards a suite of coarse-grained models for molecular simulation of monoclonal antibodies and therapeutic proteins, J Phys Chem B, 125:3574-3588 (2021)
- Woldeyes MA, Qi W, Razinkov V, Furst EM*, Roberts CJ*, Temperature-dependence of protein solution viscosity and protein-protein interactions: Insights for the origins of high-viscosity protein solutions, Mol Pharm, 17:4473-4482 (2020)
- Woldeyes MA, Calero-Rubio C, Furst EM, Roberts CJ*, Light scattering to quantify protein-protein interactions at high protein concentrations, Methods Mol Biol, 2039:23-37 (2019)
- Calero-Rubio C, Saluja A, Sahin E, Roberts CJ*, Predicting high-concentration interactions of monoclonal antibody solutions: Comparison of theoretical approaches for strongly attractive vs. repulsive conditions, J Phys Chem B, 123:5709-5720 (2019)
- Woldeyes MA, Battistoni C, Razinkov V, Furst EM*, Roberts CJ*, How well do electrostatic protein-protein interactions predict changes in viscosity of monoclonal antibody solutions?, J Pharm Sci, 108:142-154 (2019)
- Woldeyes MA, Lam LJ, Leiske D, Galush W, Roberts CJ, Furst EM, Viscosities and protein interactions of bispecific antibodies and their monospecific mixtures, Mol Pharm, 15:4745-4755 (2018)
- Calero-Rubio C, Saluja A, Ghosh R, Roberts CJ, Predicting high-concentration monoclonal antibody interactions with dilute solution data and coarse-grained molecular simulations, J Pharm Sci, 107:1269-1281 (2018)
- Woldeyes MA, Calero-Rubio C, Furst EM*, Roberts CJ*, Predicting protein interactions in concentrated globular protein solutions using colloidal models, J Phys Chem B, 121:4756-4767 (2017)
- Calero-Rubio C, Saluja A, Roberts CJ*, Coarse-grained antibody models for “weak” protein-protein interactions from low to high concentrations, 120:6592-6605 (2016)
- Ghosh R, Calero-Rubio C, Saluja A, Roberts CJ*, Relating protein-protein interactions and aggregation rates from low to high concentrations, J Pharm Sci, 105:1086-1096 (2016)
- Barnett GV, Qi W, Amin S, Lewis EN, Razinkov V, Kerwin BA, Liu Y, Roberts CJ*, Structural Changes and Aggregation Mechanisms for Anti-streptavidin IgG1 at Elevated Concentration, J Phys Chem B, 119:15150-15163 (2015)
- Barnett GV, Qi W, Amin S, Lewis EN, Roberts CJ*, Aggregate structure, morphology and the effect of aggregation mechanisms on viscosity at elevated protein concentrations, Biophys Chem, 207:21-29 (2015)
- Blanco MA, Martorana E, Manno M, Perevozchikova T, Roberts CJ*, Protein-protein interactions in dilute to concentrated solutions: alpha-chymotrypsinogen at acidic pH, J Phys Chem B, 118: 5817-5831 (2014)
- Amin S,* Barnett GV, Pathak J,* Roberts CJ, Sarangapani PS, Protein Aggregation, Particle Formation, Characterization, and Rheology, Curr Opin Coll Int Sci, 19:439-449 (2014)
Modeling and design for folding and assembly of proteins, peptides, and hybrid materials
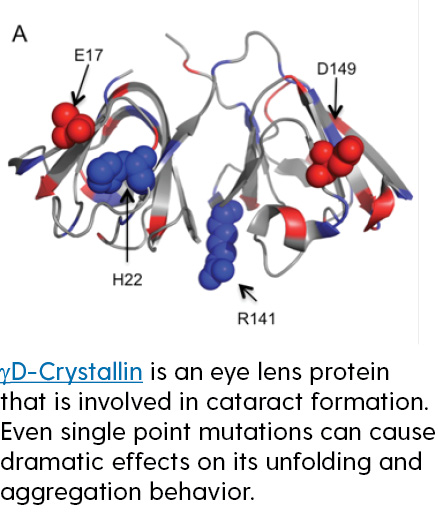 Folding and assembly of proteins requires a careful balance of a number of competing forces at the molecular level. Similarly, assembly of advanced nanostructured materials can take advantage of those competing forces. However, in both cases it is difficult to a priori predict what affects a change in the chemical structure of a protein or peptide-based material will have on the final structure and morphology of the new protein or material. Research in this area focuses on development and application of existing and new, improved models to predict how changes in variables such as peptide sequence will alter the conformational stability and aggregation of proteins, as well as the assembly of peptide-based hybrid materials.
Folding and assembly of proteins requires a careful balance of a number of competing forces at the molecular level. Similarly, assembly of advanced nanostructured materials can take advantage of those competing forces. However, in both cases it is difficult to a priori predict what affects a change in the chemical structure of a protein or peptide-based material will have on the final structure and morphology of the new protein or material. Research in this area focuses on development and application of existing and new, improved models to predict how changes in variables such as peptide sequence will alter the conformational stability and aggregation of proteins, as well as the assembly of peptide-based hybrid materials.
Recent Publications:
- Shahfar H, Du Q, Parupudi A, Shan L, Esfandiary R, Roberts CJ*, Electrostatically driven protein-protein interactions: quantitative prediction of second osmotic virial coefficients to aid antibody design, J Phys Chem Lett, 13:1366-1372 (2022)
- Meric G, Naik S, Hunter AK, Robinson AS*, Roberts CJ*, Challenges for design of aggregation-resistant variants of granulocyte-colony stimulating factor, Biophys Chem, 277:106630 (2021)
- Shahfar H**, Forder J**, Roberts CJ*, Towards a suite of coarse-grained models for molecular simulation of monoclonal antibodies and therapeutic proteins, J Phys Chem B, 125:3574-3588 (2021)
- Paik B, Calero-Rubio C, Lee J, Jia X, Kiick KL*, Roberts CJ*, Characterizing aggregate growth and morphology of alanine-rich polypeptides as a function of sequence chemistry and solution temperature from scattering, spectroscopy, and microscopy, Biophys Chem, 267:106481 (2020)
- Ferreira GM, Shahfar H, Remmele RL, Sathish H, Roberts CJ*, Identifying key amino acids that drive electrostatic attractions between therapeutic antibodies, J Phys Chem B, 123:10642-10653 (2019)
- Calero-Rubio C, Saluja A, Sahin E, Roberts CJ*, Predicting high-concentration interactions of monoclonal antibody solutions: Comparison of theoretical approaches for strongly attractive vs. repulsive conditions, J Phys Chem B, 123:5709-5720 (2019)
- O’Brien CJ, Calero-Rubio C, Razinkov V, Robinson AS, Roberts CJ, Biophysical characterization and molecular simulation of electrostatically driven self-association of an anti-fluorescein single-chain antibody, Protein Sci, 27:1275-1285 (2018)
- Meric UG, Robinson AS, Roberts CJ, Driving forces for non-native protein aggregation and approaches for predicting aggregation-prone regions, Ann Rev Chem Biomol Eng, 8 (2017), DOI: 10.1146/annurev-chembioeng-060816-101404
- Calero-Rubio C, Paik BA, Jia X, Kiick KL, Roberts CJ, Predicting unfolding thermodynamics and stable intermediates for alanine-rich helical peptides with the aid of coarse-grained molecular simulation, Biophys Chem, 217:8-19 (2016)
- O’Brien CJ, Blanco MA, Robinson AS, Roberts CJ, Modulating Non-Native Aggregation and Electrostatic Protein-Protein Interactions with Computationally Designed Single Point Mutations, Protein Engineering Design & Selection, 29:231-243 (2016)
- Paik B, Blanco MA, Roberts CJ,* Jia X, Kiick KL*, Aggregation of poly(acrylic acid)-containing elastin-mimetic copolymers, Soft Matte, 11:1839-1850 (2015)
- Paik B, Blanco MA, Roberts CJ,* Jia X, Kiick KL*, Aggregation of poly(acrylic acid)-containing elastin copolymers, Soft Matter, 11:1839-1850 (2015)
- Costanzo JA, O’Brien CJ, Tiller K, Tamargo E, Robinson AS, Roberts CJ*, Fernandez EJ*, Computational Design to Control Protein Aggregation Rates Through Conformational Stability, Protein Engineering Design & Selection, 27: 157-167 (2014)
- Blanco MA, Sahin E, Robinson AS, Roberts CJ*, Coarse-grained model of protein-protein interactions, B22, and protein cluster formation, J Phys Chem B, 117: 16013-16028 (2013)
- Grünberger A, Lai P-K, Blanco MA, Roberts CJ*, Coarse-grained modeling of protein second osmotic virial coefficients: sterics and non-specific attractions, J Phys Chem B, 117:763-770 (2013)
- Top A, Zhong S, Yan C, Roberts CJ*, Pochan DJ*, Kiick KL*, Controlling assembly of helical polypeptides via PEGylation strategies, Soft Matter, 7:9758-9766 (2011)
- Top A, Roberts CJ*, Kiick KL*, Conformational and aggregation properties of a PEGylated alanine-rich polypeptide, Biomacromolecules, 12:2184–2192 (2011)
- Sahin E, Jordan JL, Zhang A, Naranjo A, Costanzo JA, Weiss WF IV, Spatara ML, Robinson AS, Fernandez EJ*, Roberts CJ*, Computational design and biophysical characterization of point mutations for gamma-D-crystallin illustrate a balance of conformational stability and intrinsic aggregation propensity, Biochemistry, 50:628-639 (2011)
Improved prediction of protein (in)stability
Proteins degrade in a variety of ways, and must remain viable (with minimal degradation) over multiple years in order to be commercially practical as drug products. However, early-stage development activities are typically based on small supplies of protein material, and require development decisions to be reached on time scales that are much shorter than those needed to directly monitor protein degradation in the laboratory. In addition, degradation can be accelerated unexpectedly once the protein is produced in commercial-scale facilities and/or is exposed to deviations from its recommended storage conditions. Historically, proteins have required refrigerated or even frozen storage in order to maintain sufficient shelf life. However, this neglects a large portion of the world’s population that do not have the infrastructure to maintain a “cold chain” for the lifetime of a protein product. Research in the group focuses on development of improved engineering-science approaches and physics-based models to predict key product attributes such as aggregate formation, phase separation, and rheological properties.
Recent Publications:
- Shahfar H, Du Q, Parupudi A, Shan L, Esfandiary R, Roberts CJ*, Electrostatically driven protein-protein interactions: quantitative prediction of second osmotic virial coefficients to aid antibody design, J Phys Chem Lett, 13:1366-1372 (2022)
- Gomes DC, Teixeira SCM, Leao JB, Butler P, Razinkov VI, Qi W, Rodriguez MA, Roberts CJ*, In situ monitoring of protein unfolding / conformational states under cold, high-pressure stress, Mol Pharm, 18:4415-4427 (2021)
- Meric G, Naik S, Hunter AK, Robinson AS*, Roberts CJ*, Challenges for design of aggregation-resistant variants of granulocyte-colony stimulating factor, Biophys Chem, 277:106630 (2021)
- Shahfar H**, Forder J**, Roberts CJ*, Towards a suite of coarse-grained models for molecular simulation of monoclonal antibodies and therapeutic proteins, J Phys Chem B, 125:3574-3588 (2021)
- Wood CV, Qi W, Razinkov V, Furst EM*, Roberts CJ*, A rapid, small-volume approach for protein aggregation via air-water Interfaces, J Pharm Sci, 110:1083-1092 (2021)
- Wood CV, Razinkov V, Qi W, Furst EM*, Roberts CJ*, Kinetics and competing mechanisms of antibody aggregation via bulk solution and surface-mediated pathways, J Pharm Sci 109:1449-1459 (2020)
- Ferreira GM, Shahfar H, Remmele RL, Sathish H, Roberts CJ*, Identifying key amino acids that drive electrostatic attractions between therapeutic antibodies, J Phys Chem B, 123:10642-10653 (2019)
- Gomes D, Kalman RK, Pagels R, Rodrigues MA, Roberts CJ*, Parallel Chromatography and In Situ Scattering to Interrogate Competing Protein Aggregation Pathways, Protein Sci, 27:1325-1333 (2018)
- O’Brien CJ, Calero-Rubio C, Razinkov V, Robinson AS, Roberts CJ, Biophysical characterization and molecular simulation of electrostatically driven self-association of an anti-fluorescein single-chain antibody, Protein Sci, 27:1275-1285 (2018)
- Calero-Rubio C, Saluja A, Ghosh R, Roberts CJ, Predicting high-concentration monoclonal antibody interactions with dilute solution data and coarse-grained molecular simulations, J Pharm Sci, 107:1269-1281 (2018)
- Rosa MF, Roberts CJ, Rodriguez MA*, Connecting High-Temperature and Low-Temperature Protein Stability and Aggregation, PLOS One, (online only) DOI: 10/1371/journal.pone.0176748 (2017)
- Meric UG, Robinson AS*, Roberts CJ*, Driving forces for non-native protein aggregation and approaches for predicting aggregation-prone regions, Ann Rev Chem Biomol Eng, 8 (2017), DOI: 10.1146/annurev-chembioeng-060816-101404
- Barnett GV, Drenski MC, Razinkov V, Reed WF*, Roberts CJ*, Protein aggregation mechanisms and kinetics from combined monomer depletion rates and continuous scattering, Analytical Biochem, 511:80-91 (2016)
- Ghosh R, Calero-Rubio C, Saluja A, Roberts CJ*, Relating protein-protein interactions and aggregation rates from low to high concentrations, J Pharm Sci, 105:1086-1096 (2016)
- Barnett GV, Razinkov V, Kerwin BA, Hillsley A, Roberts CJ*, Acetate and Citrate Specific-Ion-Effects on Temperature-Dependent Aggregation Rates of Anti-Streptavidin IgG1, J Pharm Sci, 105:1066-1073 (2016)
- Roberts CJ*, Nesta DP, Kim N., Effects of temperature and osmolytes on parallel degradation routes for an IgG1 antibody, J Pharm Sci, 102:3556-3566 (2013)
- Wang W*, Roberts CJ*, Non-Arrhenius Protein Aggregation, AAPS J, (online only) doi:10.1208/s12248-013-9485-3 (2013)
- Kroetsch AM, Sahin E, Wang H-Y, Krizman S, Roberts CJ*, Relating particle formation to salt- and pH-dependent phase separation of non-native aggregates of alpha-chymotrypsinogen A, J Pharm Sci, 101:3651-3660 (2012)
- Brummitt RK, Andrews JM, Jordan JL, Fernandez EJ, Roberts CJ*, Thermodynamics of amyloid dissociation provide insights into aggregate stability regimes, Biophys Chem, 168-169: 10-18 (2012)
- Roberts CJ*, Das TK, Sahin E, Predicting Aggregation Rates for Therapeutic Proteins: Approaches and Challenges, Int J Pharm, 418:318-333 (2011)
- Brummitt, RK, Nesta DP, Roberts, CJ*, Predicting accelerated and low-temperature aggregation rates for monoclonal antibody formulations, J Pharm Sci, 100:4234-4243 (2011)
Protein adsorption at solid-liquid and air-liquid interfaces
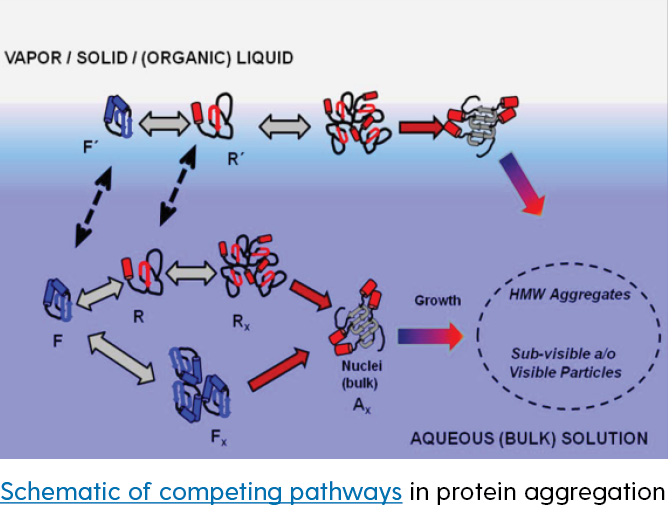 Protein adsorption to bulk interfaces is often unavoidable, and can have significant impacts on protein stability in the context of the manufacture, storage, and administration of protein-based products. Efforts in this research area focus generally on understanding key factors that control the adsorption/desorption processes, the structural nature of adsorbed protein films, how these processes impact the formation of protein aggregates and visible/subvisible particles that are potentially detrimental in protein-based products, and how best to measure properties of the interface.
Protein adsorption to bulk interfaces is often unavoidable, and can have significant impacts on protein stability in the context of the manufacture, storage, and administration of protein-based products. Efforts in this research area focus generally on understanding key factors that control the adsorption/desorption processes, the structural nature of adsorbed protein films, how these processes impact the formation of protein aggregates and visible/subvisible particles that are potentially detrimental in protein-based products, and how best to measure properties of the interface.
Recent Publications:
- Wood CV, Qi W, Razinkov V, Furst EM*, Roberts CJ*, A rapid, small-volume approach for protein aggregation via air-water Interfaces, J Pharm Sci, 110:1083-1092 (2021)
- Papachristodoulou M, Doutch J, Sang H, Leung B, Church A, Charleston T, Clifton L, Butler P, Roberts CJ, Bracewell DG*, In-situ neutron scattering of antibody adsorption during protein A chromatography, J Chromatography A 1617:460842 (2020)
- Wood CV, Razinkov V, Qi W, Furst EM*, Roberts CJ*, Kinetics and competing mechanisms of antibody aggregation via bulk solution and surface-mediated pathways, J Pharm Sci 109:1449-1459 (2020)
- Papachristodoulou M, Doutch J, Sang H, Leung B, Church A, Charleston T, Clifton L, Butler P, Roberts CJ, Bracewell DG*, In-situ neutron scattering of antibody adsorption during protein A chromatography, J Chromatography A (in press)
- Mazzer A, Nanda T, Butler P, Roberts CJ, Clifton L, Bracewell DG*, Neutron reflectivity measurement of protein A — antibody complex at the solid-liquid interface, J Chromatography A, 1499:118-131 (2017)
- Perevozchikova T, Nanda H, Nesta DP, Roberts CJ*, Protein adsorption, desorption, and aggregation mediated by solid-liquid interfaces, J Pharm Sci, 104:1946-1959 (2015)
- Roberts, CJ Nucleation, Aggregation, and Conformational Distortion, Ch. 5 in Biophysical Methods for Biotherapeutics: Discovery and Development Applications, Das, TK (Ed.), John Wiley & Sons, Hoboken, NJ, 2014
